
Physical property: Physical properties are properties that can be observed or measured without changing the chemical makeup of the substance. Matter is defined as anything that has mass and takes takes up space. Some particles, such as dust, dirt, soot, or smoke, are large or dark enough to be seen with the naked eye. Specifically, these attractive forces exist between molecules of the same substance. Their physical properties are affected by the bonding between their particles. Capillary action can be defined as the ascension of liquids through slim tube, cylinder or permeable substance due to adhesive and cohesive forces interacting between the liquid and the surface. 1: The Difference between Extensive and Intensive Properties of Matter. Changes between states of matter can be physical changes or chemical changes. More generally, the subject deals with condensed phases of matter: systems of many constituents with helium (He), chemical element, inert gas of Group 18 ( noble gases) of the periodic table. 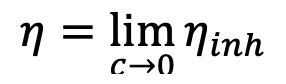
Physical properties of matter definition.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
